Cellular Conversations: Signal-Driven Regeneration – Advances in Orthopedic Healing, Electrical Stimulation
In our Cellular Conversations blog post series, we described the role of biochemical, mechanical, and electrical signals in bone tissue repair.
Immune System and Bone Healing: Understanding the Connection

Healing a broken bone is no small feat and requires a highly sophisticated, coordinated series of interactions, many of which are orchestrated by the immune system.
From Infection to Inflammation: How Immunity Shapes Bone Health

In this second installment of our blog series on Osteoimmunology, we’ll explore the intricate connection between immunity and bone health, and how this interplay influences repair processes.
Cellular Conversations: Signal-Driven Regeneration – Advances in Orthopedic Healing, Biochemical and Mechanical Stimulation

In our Cellular Conversations blog post series, we described the biochemical, mechanical and electrical signals that bone cells use to facilitate growth and repair. This post delves into how scientists and surgeons are leveraging the knowledge of biochemical and mechanical signals to develop advanced bone repair therapies.PART 3: Biochemical Signals – Enhancing Repair with Growth Factors and CytokinesOf the many growth factors explored for bone repair, bone morphogenetic proteins have received the most attention in terms of research, FDA-approvals, and clinical use (Table 1).Table 1: Clinical Use of Biochemical Signals for Bone Repair Edit Growth Factor Role Application Products Bone Morphogenetic Proteins (BMPs)1-8 Differentiation of mesenchymal stem cells (MSC) into osteoblasts Induction of osteogenesis Regulation of chondrogenesis Spinal fusions, tibial fractures, cranioplasty BMP-2 (rhBMP-2) BMP-7 (OP-1) FDA-approved 2002 Platelet-Derived Growth Factor (PDGF)6,8 Stimulates cell proliferation, angiogenesis, and recruitment of MSC to the repair site Periodontal bone repair, surgical fusion of the ankle and hindfoot, distal radius fractures. GEM 21S FDA-approved 2005 Vascular Endothelial Growth Factor (VEGF)6 Improves repair site vascularization Ossification and vascularization in critical-sized mandibular bone and calvaria defects. Not approved stand-alone Fibroblast Growth Factor-2 (FGF-2)6,9 Stimulates angiogenesis Proliferation of osteogenic cells Regeneration of mandible cortical bone Approved in some countries Insulin-like Growth Factor-1 (IGF-1)10,11 Enhances osteoblast proliferation and differentiation Matrix synthesis Clinical studies to improve bone healing with growth hormone Not approved stand-alone Parathyroid Hormone (PTH)12 Stimulates osteoblast activity and bone remodeling Osteoporosis treatment, off-label use for bone healing Teriparatide (PTH 1-34) Therapeutic use of growth factors presents challenges, including high production costs and the need for controlled application to avoid complications like ectopic bone formation, inflammation, or increased cancer risks. Novel carriers and biologics such as polymers, composites, hydrogels, ceramics, and others are under study to provide controlled and sustained release methods. 2,3,5,7 Mechanical Signals – Promoting Faster Bone Repair (Table 2)The most common clinical use of mechanical signaling in bone repair is through physical therapy, although results are difficult to quantify. Physical therapy plays a crucial role in the mechanical stimulation of bone growth through weight-bearing exercises, which apply load stress to the bone, promoting remodeling and growth. Range of motion exercises enhance blood flow and nutrient delivery to the fracture site, while muscle strengthening exercises increase support and stability, effectively distributing the mechanical load. These combined methods promote bone mass and mineralization; however, standardizing these therapies can be challenging due to factors like fracture location and treatment variables.Table 2: Clinical Use of Mechanical Signals for Bone Repair Edit Mechanical Signal Application Advantages Limitations Distraction Osteogenesis13 Oral, orthopedic, craniofacial, and plastic surgery No bone tissue transplant needed Long consolidation period, pain, infection, nonunion Physical Therapy14 Enhances fracture healing Prevents bone loss Promotes bone mass and remodeling Fatigue, lack of motivation, long duration required Low-Intensity Pulsed Ultrasound (LIPUS)15 Stimulates bone formation and healing at fracture sites Noninvasive, no side effects, low cost No improvement in weight-bearing capacity, pain reduction External Fixators16 Stabilize fractures Aid weight-bearing to promote healing Provides stability, corrects alignment Long duration for frame removal, pin-track infections Other techniques such as distraction osteogenesis and low-intensity pulsed ultrasound (LIPUS) are also used clinically to improve bone repair through mechanical signaling. Distraction osteogenesis is a surgical technique involving an incision in the bone to place a distractor, which gradually separates the bone at a controlled rate, typically a few millimeters per day. As the bone is stretched, new tissue forms within the gap, allowing for bone lengthening or repositioning. This technique is used in craniofacial surgery to correct jaw deformities and in trauma-related orthopedic surgeries but is associated with a high risk of complications.17Despite FDA approval for nonunion fractures, low-intensity pulsed ultrasound (LIPUS) products have not consistently reduced recurrent fractures.14-16 External fixators aid in controlled movement and load distribution across the fracture site, essential for bone healing. Pins secure the external frame of clamps and rods to the bone, but careful management is required to mitigate the risk of infection.At Molecular Matrix, Inc., we leverage our expertise in signal-driven regeneration to develop novel bone graft substitutes, facilitating effective treatment for fractures and bone injuries. To learn more about Molecular Matrix, Inc. click here.References Singh H, Moss IL. Biologics in Spinal Fusion. In: Biologics in Orthopaedic Surgery [Internet]. Elsevier; 2019 [cited 2024 Jun 20]. p. 165–74. Available from: https://linkinghub.elsevier.com/retrieve/pii/B9780323551403000151 Senarath-Yapa K, McArdle A, Renda A, Longaker M, Quarto N. Adipose-Derived Stem Cells: A Review of Signaling Networks Governing Cell Fate and Regenerative Potential in the Context of Craniofacial and Long Bone Skeletal Repair. Int J Mol Sci. 2014 May 26;15(6):9314–30. Mariani E, Pulsatelli L, Facchini A. Signaling Pathways in Cartilage Repair. Int J Mol Sci. 2014 May 15;15(5):8667–98. Halloran D, Durbano HW, Nohe A. Bone Morphogenetic Protein-2 in Development and Bone Homeostasis. J Dev Biol. 2020 Sep 13;8(3):19. Mendenhall SK, Priddy BH, Mobasser JP, Potts EA. Safety and efficacy of low-dose rhBMP-2 use for anterior cervical fusion. Neurosurg Focus. 2021 Jun;50(6):E2. Oliveira ÉR, Nie L, Podstawczyk D, Allahbakhsh A, Ratnayake J, Brasil DL, et al. Advances in Growth Factor Delivery for Bone Tissue Engineering. Int J Mol Sci. 2021 Jan 18;22(2):903. Tateiwa D, Kaito T. Advances in bone regeneration with growth factors for spinal fusion: A literature review. North Am Spine Soc J NASSJ. 2023 Mar;13:100193. Gillman CE, Jayasuriya AC. FDA-approved bone grafts and bone graft substitute devices in bone regeneration. Mater Sci Eng C. 2021 Nov;130:112466. Krticka M, Planka L, Vojtova L, Nekuda V, Stastny P, Sedlacek R, et al. Lumbar Interbody Fusion Conducted on a Porcine Model with a Bioresorbable Ceramic/Biopolymer Hybrid Implant Enriched with Hyperstable Fibroblast Growth Factor 2. Biomedicines. 2021 Jun 25;9(7):733. Giustina A, Mazziotti G, Canalis E. Growth Hormone, Insulin-Like Growth Factors, and the Skeleton. Endocr Rev. 2008 Aug 1;29(5):535–59. Locatelli V, Bianchi VE. Effect of GH/IGF-1 on Bone Metabolism and Osteoporsosis. Int J Endocrinol. 2014;1–25. Silva BC, Bilezikian JP. Parathyroid hormone: anabolic and catabolic actions on the skeleton. Curr Opin Pharmacol. 2015 Jun;22:41–50. Yang S, Wang N, Ma Y, Guo S, Guo S, Sun H. Immunomodulatory effects and mechanisms of distraction osteogenesis. Int
Cellular Conversations: Biological Signals in Orthopedic Tissue Repair
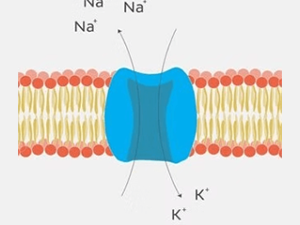
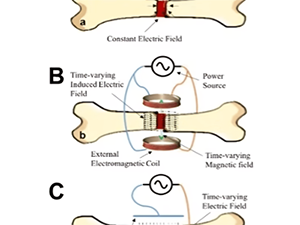
Part 3: Electrical Signals Cellular communication is crucial in processes like cell differentiation, growth, development, tissue and organ formation, and physiological regulation. In Part 1 and Part 2 of this blog series, we focused on biochemical and mechanical signaling in bone cells. Now, we explore another fascinating aspect of cellular communication: electrical signals and their influence on orthopedic tissue repair. Bone ElectrophysiologyThe electrical potential of bone is a intriguing aspect of its physiology. Let’s first explore terms used to describe electrical potentials involving cells and tissues, including bone:Dielectric: The ability to store and dissipate electrical energy. The cell membrane, composed of a lipid bilayer, typically acts as an electrical insulator but can be polarized in the presence of an electrical force, contributing to its dielectric behavior.Piezoelectric: Electric charge produced in response to mechanical stress, primarily attributed to the crystal structure of collagen molecules in the bone.Pyroelectric: The ability of bone to generate an electric charge in response to temperature changes.Ferroelectric: Spontaneous electric polarization which is reversible with the application of an external electric field.These potentials work together with physiological ion movements to produce interactive crosstalk between the organic and inorganic components of bone and between bone cells and their extracellular matrix, playing roles in bone metabolism, homeostasis, and regeneration.Electroactive and Electrosensitive Constituents of Bone CellsCellular Transmembrane Potential: Differences in sodium and potassium ion concentrations create a transmembrane electrical field in bone cells, altering cell metabolism and signaling pathways controlling bone tissue homeostasis and remodeling.Voltage-Sensitive Ion Channels: Sodium, potassium, calcium, and chloride ion channels are present in bone cells and respond to electrical stimuli with ion flux that affects bone maintenance, deposition, and resorption.Intracellular Signaling Pathways: Electrical stimuli may activate or inhibit intracellular signaling pathways which, in turn, influence downstream gene and protein expression.Extracellular Matrix Proteins: Structural characteristics of the bone’s extracellular matrix enhance its electrical properties, contributing to its dielectric, piezoelectric, pyroelectric, and ferroelectric properties.Proteoglycans and Glycosaminoglycans: As we discussed in a previous post, glycosaminoglycans are polysaccharides with chains of repeating units. When attached to a protein core, they form proteoglycans. Through the action of anionic (negatively charged) glycosaminoglycan chains these molecules generate streaming potentials, influencing osteocyte activity and bone formation or resorption.Hydroxyapatite: Influences bone’s bioelectrical properties by restricting the hydrogen bonding of collagen fibers with water molecules, influencing collagen fiber orientation and mechanical response to compressive forces. Hydration status plays a role here too, as water-saturated collagen fibers align more symmetrically, short-circuiting the electrical potential.Osteocytes, the most abundant cells in bone tissue, play a crucial role in maintaining bone health. They are embedded within the mineralized bone matrix and have long, dendritic-like cell processes that extend through the canaliculi to form gap junctions with adjacent osteocytes and osteoblasts. The result is a functional syncytium of interconnected cells throughout bone tissue that acts to transmit electrical and other signals.One of the key mechanisms by which bone cells communicate electrically is through mechanotransduction. This process involves the conversion of mechanical stimuli, such as strain or pressure, into electrical signals. When bone tissue is subjected to mechanical stress, it generates electrical potentials known as streaming potentials. These potentials are detected by osteocytes, then relayed to other bone cells via the molecules and mechanisms discussed above.Electrical communication plays a crucial role in bone metabolism, homeostasis, and regeneration. The dielectric, piezoelectric, pyroelectric, and ferroelectric properties of bone enable response to various stimuli and coordination of cellular activities. The electroactive constituents of bone cells, including transmembrane potentials and various channels and proteins, transmit and/or enhance this communication. As research continues to uncover the intricacies of electrical signaling in bone cells, new therapeutic approaches for orthopedic tissue repair and bone-related disorders may emerge.At Molecular Matrix, Inc., we work to understand these mechanisms and explore exciting possibilities for improving bone health and the development of innovative treatments for bone repair. To learn more about Molecular Matrix, Inc.; visit www.molecularmatrix.com.Look for our next post where we will discuss the application of electrical signaling in bone healing therapeutics!References: deVet, T., Jhirad, A., Pravato, L., & Wohl, G. R. (2021). Bone Bioelectricity and Bone-Cell Response to Electrical Stimulation: A Review. Critical Reviews in Biomedical Engineering, 49(1), 1–19. https://doi.org/10.1615/CritRevBiomedEng.2021035327 Heng, B. C., Bai, Y., Li, X., Meng, Y., Lu, Y., Zhang, X., & Deng, X. (2023). The bioelectrical properties of bone tissue. Animal Models and Experimental Medicine, 6(2), 120–130. https://doi.org/10.1002/ame2.12300 Sun, J., Xie, W., Wu, Y., Li, Z., & Li, Y. (2024). Accelerated Bone Healing via Electrical Stimulation. Advanced Science, 2404190. https://doi.org/10.1002/advs.202404190
Cellular Conversations: Biological Signals in Orthopedic Tissue Repair
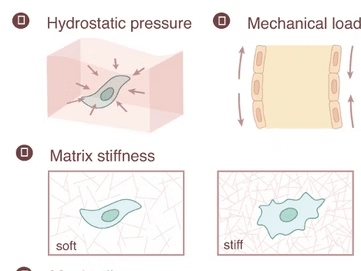
Part 2: Mechanical SignalsImagine our cells communicating like a vast network of people. While we use words, cells “talk” through biochemical, mechanical, and electrical signals. At Molecular Matrix, Inc., we’re dedicated to unraveling these cellular conversations to improve regenerative therapies. In this series, we explore key biological signals involved in orthopedic tissue repair. In Part 1, we covered biochemical signals crucial for bone repair. Now, in Part 2, we dive into the role of mechanical signals and how they guide bone healing.Mechanical Signals: How Physical Forces Stimulate Bone HealingOur bones endure continuous stress, from simple movements to intense physical activities. Over a century ago, Wolff’s Law described how bones adapt to the level of mechanical load: increased load strengthens bone, while lack of load leads to weakening. This concept underlies recovery strategies post-injury, injury prevention, and management of conditions such as osteoporosis through regular resistance training or weight-bearing exercises.How Cells Sense and Respond to Mechanical Stimuli Cells interpret mechanical signals through a process called mechanotransduction, where physical forces transform stimuli into biochemical cues that drive bone remodeling. Various cellular receptors and molecules help convert these forces into signals that promote growth, repair, or adaptation in response to stress. (Robling et al., 2006)Here is a closer look at the types of bone cells involved in responding to these signals: Osteocytes: These mature bone cells are nestled in the mineralized matrix, connected by channels that sense fluid shifts during movement. Changes in fluid shear stress prompt osteocytes to signal other cells to either build bone when under load stress or increase bone resorption when unstressed. (Qin et al., 2020; Xu et al., 2021) Mesenchymal Stem Cells (MSCs): Fluid shear stress encourages MSCs to differentiate into osteoblasts and suppresses the formation of osteoclasts. (Datta et al., 2006; Henstock et al., 2024) Osteoclasts: These bone-resorbing cells have ion channels sensitive to fluid stress that regulate their activity, morphology, and gene expression. (Bratengeier et al., 2020) Osteoblasts: These bone-building cells respond to physical load by producing proteins and growth factors that promote bone matrix formation and stabilization at injury sites. (Knapik et al., 2014) The Extracellular Matrix (ECM) and MechanotransductionAll the important cellular players in bone physiology are influenced by the mechanical properties of the three-dimensional environment in which they reside. This ‘niche’ consists of ECM components together with the cells they surround and connect. The ECM itself is composed of proteins like collagen, fibronectin, elastin, laminin, glycosaminoglycan, and glycoproteins. The specific composition of the ECM is dynamic with changes in density, rigidity and orientation occurring in response to the extracellular environment. This, in turn, may induce reorganization of cytoskeletal protein and trigger changes in downstream molecular signaling pathways. As examples: MSC differentiation can be switched from adipogenesis to osteogenesis by changing ECM stiffness. Osteoblast maturation increases on firm ECM while soft ECM appears to favor osteoclast maturation. Mechanosensitive Cell Structures Focal adhesions: These large macromolecular protein complexes can contain over 100 different proteins and have roles in cell migration. Integrins are a crucial transmembrane component of focal adhesions. These heterodimeric receptors functionally link the ECM to the cytoskeleton, providing a means to transfer mechanical stimuli from the extracellular environment to intracellular signaling or cytoskeletal proteins. Primary Cilium: Mechanical signals such as fluid shear stress are detected by cilium and transmitted via the hedgehog signaling pathway. In bone, this signals osteoprogenitors to differentiate into bone-forming osteoblasts at certain frequencies. G protein-coupled receptors: These receptors, located on the cell membrane, sense mechanical stimuli and transduce the signal via the Rho-Rock and PLC-IP3 signaling pathways. (Kalinkovich & Livshits, 2018) Stretch-Activated Ion channels: Ion flow, especially calcium influx, modulates downstream signaling. (Martino et al., 2018) Cytoskeleton: Within the cell is a dynamic network of interlinking protein filaments and tubules known as the cytoskeleton. In addition to providing for cell shape, organization of organelles and cell movement, the cytoskeleton provides a pathway for mechanical forces to be transferred from the plasma membrane to internal cellular structures. (Maniotis et al., 1997) Enhancing Bone Healing Through Controlled Mechanical Load Controlled physical load during rehabilitation can stimulate bone healing by strengthening the initial callus and promoting complete remodeling. Advanced biomaterials and implants are being developed to mimic mechanical cues, helping guide cell behavior and optimize healing. (Hart et al., 2017) Mechanical signals are a crucial component of the body’s repair toolkit. Understanding and leveraging these forces could enable us to better harness the healing potential of orthopedic tissues. At Molecular Matrix, Inc., our bone graft substitute Osteo-P® BGS models the natural bone extracellular matrix, providing substrate and mechanical cues for repair with the patient’s own cells. Through these cellular conversations, we’re developing new possibilities in regenerative medicine that are both innovative and aligned with the body’s intrinsic healing abilities. References: Bratengeier, C., Liszka, A., Hoffman, J., Bakker, A. D., & Fahlgren, A. (2020). High shear stress amplitude in combination with prolonged stimulus duration determine induction of osteoclast formation by hematopoietic progenitor cells. FASEB journal : official publication of the Federation of American Societies for Experimental Biology, 34(3), 3755–3772. https://doi.org/10.1096/fj.201901458R Datta, N., Pham, Q. P., Sharma, U., Sikavitsas, V. I., Jansen, J. A., & Mikos, A. G. (2006). In vitro generated extracellular matrix and fluid shear stress synergistically enhance 3D osteoblastic differentiation. Proceedings of the National Academy of Sciences of the United States of America, 103(8), 2488–2493. https://doi.org/10.1073/pnas.0505661103 Hart NH, Nimphius S, Rantalainen T, Ireland A, Siafarikas A, Newton RU. Mechanical basis of bone strength: influence of bone material, bone structure and muscle action. J Musculoskelet Neuronal Interact. 2017 Sep 1;17(3):114-139. PMID: 28860414; PMCID: PMC5601257. Henstock, J. R., Price, J. C. F. A., & El Haj, A. J. (2024). Determining Which Hydrostatic Pressure Regimes Promote Osteogenesis in Human Mesenchymal Stem Cells. Tissue engineering and regenerative medicine, 10.1007/s13770-024-00666-w. Advance online publication. https://doi.org/10.1007/s13770-024-00666-w Kalinkovich, A., & Livshits, G. (2021). Biased and allosteric modulation of bone cell-expressing G protein-coupled receptors as a novel approach to osteoporosis therapy. Pharmacological research, 171, 105794. https://doi.org/10.1016/j.phrs.2021.105794 Knapik, D. M., Perera, P., Nam, J., Blazek, A. D., Rath, B., Leblebicioglu, B., Das, H., Wu, L. C., Hewett, T. E., Agarwal, S. K., Jr, Robling, A. G., Flanigan, D. C., Lee, B. S., & Agarwal, S. (2014). Mechanosignaling in bone health, trauma and inflammation. Antioxidants & redox signaling, 20(6), 970–985. https://doi.org/10.1089/ars.2013.5467 Maniotis, A. J., Chen, C. S., & Ingber, D. E. (1997). Demonstration of mechanical connections between integrins, cytoskeletal filaments, and
Cellular Conversations: Biological Signals in Orthopedic Tissue Repair
Part 1: Biochemical Signals Think of the ways we communicate daily – speaking, texting, calling, emailing. Now, imagine the human body as a vast network where cells also “talk” constantly. Though cells don’t use words, their “language” of biochemical, mechanical, and electrical signals orchestrates essential tissue repair and regeneration processes. At Molecular Matrix, Inc., we believe that understanding how these important cellular conversations influence orthopedic tissue repair will lead development of better regenerative therapies. Biological Signals: The Healing Language of Molecules Biological signals are molecular instructions that guide critical processes like inflammation, cell migration, and tissue reconstruction. In orthopedic repair, they play vital roles in activating bone and cartilage repair, modulating inflammation, and coordinating various cell types. The key biological signals in orthopedic healing can typically be grouped into three categories: biochemical, mechanical, or electrical. This blog post, the first in a three-part series, delves into biochemical signals and sets the stage for a deeper exploration of each signal group. Growth Factors: The Master Controllers of Healing Growth factors are a group of proteins that stimulate cell growth, proliferation, and differentiation. The following growth factors play a crucial role in bone repair: Bone Morphogenetic Proteins (BMPs): Among the most powerful growth factors for bone healing, BMPs are crucial in signaling mesenchymal stem cells (MSC) to differentiate into osteoblasts. BMP-2, for example, is FDA-approved for complex fracture and spinal fusion surgeries, proving its critical role in clinical bone repair. (Dumic-Cule et al., 2018) Transforming Growth Factor-Beta (TGF-β): Essential for both inflammation control and cartilage formation, TGF-β promotes chondrocyte formation and helps regenerate damaged cartilage, making it crucial in joint and cartilage repair. (Wu et al., 2023) Vascular Endothelial Growth Factor (VEGF): By stimulating blood vessel formation (angiogenesis), VEGF enhances nutrient delivery to healing tissues, supporting sustained tissue repair. (Maruyama et al., 2020) Fibroblast Growth Factor (FGF): FGFs, especially FGF-2, are involved in cell proliferation and angiogenesis. FGF2 is important in tendon-to-bone healing, and repair of bone, cartilage, and tendons. In addition to acting as a mitotic promoter, FGF2 accelerates revascularization of the injured area, upregulates osteogenic and chondrogenic gene expression, and recruits and guides the migration of MSC. (Zhang et al., 2020) Wnt proteins: The Wnt signaling pathway regulates many aspects of osteoblast physiology including proliferation, differentiation, matrix formation and mineralization, and apoptosis. Wnt proteins also activate osteoclasts which help shape and remodel bone during the final stage of healing. (Bodine and Komm, 2006) Cytokines: Conductors of InflammationCytokines are small proteins that mediate inflammation, essential for tissue repair. Interleukin-1 beta (IL-1β) and Tumor Necrosis Factor-Alpha (TNF-α): These cytokines initiate inflammation through macrophage activation to facilitate phagocytosis of microbes and damaged tissue. However, they require precise regulation – excessive inflammation driven by their release of pro-inflammatory mediators can impair healing and activate osteoclasts for bone resorption. (Fullerton and Gillroy, 2016; Loi et al., 2016) Interleukin-6 (IL-6): This cytokine is produced by many cell types and binds to both membrane-bound and soluble LDL-related protein receptors. IL-6 induces and/or activates osteoclast differentiation while inhibiting osteoblast activity or differentiation. (Takeuchi et al., 2021) Chemokines: Recruiting Cells to the Repair SiteChemokines act as the “job site recruiters”, attracting stem cells and immune cells to injured tissues. CCL2: This powerful chemokine attracts MSC to the injury site and promotes crosstalk with macrophages and endothelial cells. (Shinohara et al., 2023) SDF-1 and CXCR4 axis: Ischemia induces expression of SDF-1 (stromal cell-derived factor-1) which binds to the CXCR4 receptor expressed on MSC. The SDF-1/CXCR4 signaling axis is crucial in recruiting MSCs to the ischemic injury site. Inhibition of SDF-1 or blocking of CXCR4 has been shown to prevent MSC recruitment and results in impaired bone healing. (Kitaori et al., 2009; Hermann et al., 2015) Extracellular Matrix (ECM) Proteins: Key Team Members in the Healing ProcessThe ECM, a network of proteins and molecules surrounding cells, is more than just structural support; it dynamically influences cell behavior during bone repair by releasing biochemical cues that direct cells to grow, migrate and differentiate. (Lin et al., 2020) Look for more on this topic in future blog posts. Fibronectin: Guides the chemotactic recruitment, migration and adhesion of MSC and endothelial cells by sequestering growth factors at the injury site. (Klavert and van der Eerden, 2021). Proteoglycans (link to previous post on proteoglycans): Sequester growth factors and promote collagen fibrillogenesis and mineralization. Glycoproteins: Regulate calcium release and bone remodeling. Coordinating the Process: Timing and BalanceWhile each biochemical signal is crucial to bone repair, timing and balance are key to successful healing. For instance, growth factors like BMPs and TGF-β must appear in specific sequences to guide MSCs from initial recruitment through differentiation. Similarly, cytokines help control the immune response and inflammation but must taper off to prevent excessive tissue damage.Disruption in any stage or imbalance in signaling can lead to complications, such as delayed union (where the bone heals slowly) or non-union (where the bone fails to heal). Conditions like diabetes, for instance, can disrupt normal signaling pathways, leading to compromised healing. For this reason, researchers are investigating ways to modulate these signals to enhance bone repair, especially in people with impaired healing abilities.Future Directions: Enhancing Bone RepairUnderstanding biochemical signals has led to new therapeutic strategies for bone repair. One example is the use of BMP-based bone grafts to promote healing in complex fractures. Other promising research involves gene therapy techniques aimed at activating specific signaling pathways to accelerate healing.ConclusionThe body’s process of repairing bone is a remarkable orchestration of biochemical signals, each playing a specific role to ensure that bones heal correctly and regain strength. At Molecular Matrix, Inc., we are using our understanding of these biochemical signals to develop more effective treatments for fractures and bone injuries, helping more patients recover faster and completely. (Kim and Lee, 2023) These innovative therapies will change the landscape of regenerative medicine and help individuals with chronic conditions or severe injuries regain full mobility and quality of life. To learn more, visit www.molecularmatrix.comReferences: Bodine PV, Komm BS. Wnt signaling and osteoblastogenesis. Rev Endocr Metab Disord. 2006 Jun;7(1-2):33-9. doi: 10.1007/s11154-006-9002-4. PMID: 16960757.
Bone repair and regeneration: The Immune Component Is A Healing Partner
When most people think about bone repair, they imagine a purely mechanical process: cells depositing new bone material to fix a fracture, like a bricklayer mending a broken wall. However, the reality is far more complex. One of the most surprising partners in this process is the immune system. While often associated with fighting infections, the immune system plays an indispensable role in bone repair and regeneration. Understanding this connection opens the door to new therapeutic approaches for treating bone injuries and conditions that impair healing. Osteoimmunology: The Bone-Immune ConnectionBone and immune cells share a deep, interconnected relationship. They originate from common progenitor cells, co-exist in the bone marrow cavity, and communicate via shared regulatory and signaling molecules. Bone cells influence immune cell differentiation, while immune cells regulate bone homeostasis. The cross-talk between the skeletal and immune systems is known as osteoimmunology. (Schlundt et al., 2015; Yang & Liu, 2021)The Bone Repair Process: A Quick OverviewBefore diving into the immune system’s role, it is important to understand the stages of bone healing: Inflammation: Immediately after a fracture, blood vessels are disrupted, forming a hematoma rich in immune cells, which kickstarts the healing process. Soft Callus Formation: Within day, cells from the periosteum form a soft cartilage-like tissue, called a callus, which stabilizes the fracture. Hard Callus Formation: Over time, the soft callus is replaced by hard bone, driven by osteoblasts. Bone Remodeling: The new bone is remodeled to restore the bone’s original shape and strength. The Immune System’s Role in Each Stage The Inflammatory Phase: The initial inflammatory phase is crucial, with immune cells flooding the fracture site. Macrophages, neutrophils, and lymphocytes do more than just clear the debris; they release cytokines and growth factors that coordinate the healing process. Neutrophils: These first responders initiate an acute inflammatory response, attack microbes, and regulate bone homeostasis. Macrophages: Key players in inflammation regulation and tissue regeneration, macrophages exist in two functional states: pro-inflammatory (M1) and anti-inflammatory (M2). Initially, M1 macrophages dominate, clearing necrotic tissue and secreting cytokines like TNF-α, IL-1β, and IL-6, which recruit cells needed for repair such as osteoblasts and mesenchymal stem cells (MSC). They also stimulate angiogenesis by releasing VEGF. T Cells: CD4+ T helper cells release interferon-γ (INFγ) and TNF-α which recruit MSC and immune cells to the fracture site and promote MSC differentiation into osteoblasts. (Maruyama et al., 2020; Schlundt et al., 2015; Yang & Liu, 2021) Callus Formation: As the bone repair progresses, a transition from M1 to M2 macrophages occurs. The timing of this transition is critical for the resolution of inflammation and the progression from damage control to tissue regeneration. M2 macrophages release anti-inflammatory cytokines like IL-19 and TGF-β, which suppress inflammation and promote callus formation. M2 macrophages: These cells play a critical role in promoting MSC differentiation into chondrocytes, which form the soft callus. They also support osteoblast activity, transforming the callus into hard bone, and secrete factors like BMP-2, BMP-4, and TGF-β1, which drive osteoblast proliferation. (Clark et al., 2017; Schlundt et al., 2015; Yang & Liu, 2021) Angiogenesis: M1 macrophages initiate blood vessel formation through VEGF, while M2 macrophages promote angiogenesis via MMP9 and VEGF. (Schlundt et al., 2015) Regulatory T cells (Tregs): Tregs help resolve inflammation by releasing IL-10 and TGF-β and regulate osteoclast activity through cytotoxic T-lymphocyte associated protein 4 (CTLA4), ensuring proper bone regeneration. They also inhibit CD4+ T cells to block excessive secretion of IFN-γ and TNF-α, facilitating MSC-based bone healing. CD8+ T cells: These cells release RANKL to facilitate osteoclast recruitment and activity, which aids in the removal of the fibrin thrombus during callus formation. B cells: B cells produce osteoprotegerin, which inhibits RANKL, regulating osteoclast differentiation and promoting bone formation. Bone Remodeling Phase: In this final phase, immune cells continue to regulate the balance between bone resorption and formation, ensuring the repaired bone gains strength. M1 and M2 Macrophages: The interplay between M1 and M2 macrophages is crucial in this phase. M1 macrophages promote osteoclastogenesis and bone resorption via RANKL and TNF-α, while M2 macrophages support bone formation by secreting TGF-β and VEGF. Osteoblasts: These cells, along with immune cells, secrete osteoprotegerin which inhibits RANKL, thereby preventing excessive osteoclast activation and ensuring proper balance between bone resorption and formation. (Baht et al., 2018; Schlundt et al., 2015) The Promise of Immunomodulation in Bone Repair As researchers uncover more about the intricate connections between the immune system and bone healing, new therapeutic possibilities are emerging. Targeting immune pathways could enhance bone repair in patients with fractures and/or bone-related diseases. For patients with compromised immune systems, such as those with autoimmune diseases or undergoing chemotherapy, specialized treatments that support the immune components of bone healing may be particularly beneficial. By leveraging active collaboration between the skeletal and immune systems, future therapies and biomaterials could be designed to improve the bone healing process, especially for vulnerable patients. ConclusionThe immune system is far more than just a defender against infections – it is a key partner in bone repair. Through precise coordination of inflammation, immune cells regulate the balance between bone breakdown and formation, ensuring proper healing. At Molecular Matrix, Inc., we are developing bone repair therapeutics informed by osteoimmunology, offering faster and more effective recovery solutions for patients with fractures and bone diseases.References: Baht, G. S., Vi, L., & Alman, B. A. (2018). The Role of the Immune Cells in Fracture Healing. Current Osteoporosis Reports, 16(2), 138–145. https://doi.org/10.1007/s11914-018-0423-2 Clark, D., Nakamura, M., Miclau, T., & Marcucio, R. (2017). Effects of Aging on Fracture Healing. Current Osteoporosis Reports, 15(6), 601–608. https://doi.org/10.1007/s11914-017-0413-9 Maruyama, M., Rhee, C., Utsunomiya, T., Zhang, N., Ueno, M., Yao, Z., & Goodman, S. B. (2020). Modulation of the Inflammatory Response and Bone Healing. Frontiers in Endocrinology, 11, 386. https://doi.org/10.3389/fendo.2020.00386 Pajarinen J, Lin T, Gibon E, Kohno Y, Maruyama M, Nathan K, Lu L, Yao Z, Goodman SB. Mesenchymal stem cell-macrophage crosstalk and bone healing. Biomaterials. 2019 Mar;196:80-89. doi: 10.1016/j.biomaterials.2017.12.025. Epub 2018 Jan 2. PMID: 29329642; PMCID: PMC6028312. Schlundt, C., Schell, H., Goodman, S. B., Vunjak-Novakovic, G., Duda, G. N., & Schmidt-Bleek, K. (2015).
Polysaccharides: Crafting the Perfect Microenvironment for Bone Repair
When it comes to healing broken bones, the human body is truly remarkable.
Rebuilding Better Bone: 8 Reasons Why the Bone Microenvironment is Essential
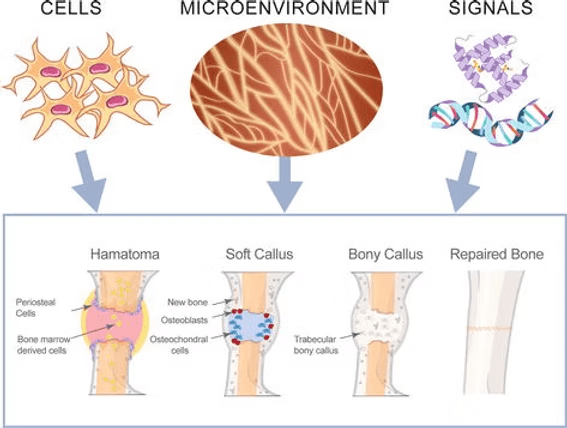
In the simplest sense, the bone microenvironment consists of the cells, molecular signals, and matrix components that create the niche for cell growth, differentiation, behavior and function.
