BMP-2 in Practice: From Bone Healing to Systemic Signaling

Bone morphogenetic proteins (BMPs) are a group of growth factors belonging to the transforming growth factor-beta (TGF-β) superfamily. First identified by Dr. Marshall Urist in 1965 for their ability to induce ectopic bone formation, BMPs have since emerged as key regulators of skeletal development and repair. BMP-2 was the first to be characterized and remains one of the most extensively studied due to its potent osteoinductive properties. Although the name BMP is descriptive of the bone-forming function of these proteins, it is quite misleading as BMPs play important roles in diverse cell types. Classification and Clinical Relevance To date, approximately 43 BMPs have been identified and categorized based on their amino acid sequence homology and biological functions: BMP Subgroup Key Functions BMP 2/4 Osteogenesis, embryogenesis Vascular development and endothelial function Inhibition of neuronal growth and differentiation BMP 5/6/7/8 Roles in bone and cartilage development Osteoblast differentiation Kidney development and repair BMP 9/10 Vascular development Bone formation BMP 12/13/14 Bone, tendon, and ligament development Among these, BMP-2 and BMP-7 have FDA approval as recombinant proteins (rhBMP-2, rhBMP-7) for use in orthopedic procedures such as spinal fusion, non-union fracture repair, and maxillofacial reconstruction [1-3]. Structural Biology of BMP-2 In general, BMPs are composed of two polypeptide chains linked by a disulfide bond, forming a dimeric molecule. BMP-2 is synthesized as a 453-residue proprotein composed of: Signal peptide: Cleaved during processing Precursor domain: Undergoes glycosylation and dimerization Mature peptide: Bioactive region formed via proteolytic cleavage from the carboxy-terminal region The mature BMP-2 exists as homodimers or heterodimers stabilized by disulfide bonds. Each monomer contains 114 amino acid residues [1,2]. Functional Roles of BMP-2 BMP-2 is synthesized in a variety of cells and tissues, typically throughout the body, including the liver, lungs, and bone (specifically in osteoblasts and osteocytes). Moreover, BMP-2 may be autocrine or paracrine, acting locally or systemically to initiate cell-to-cell responses or travel via the bloodstream to target cells [1]. BMP-2 acts as a multifunctional signaling molecule involved in: Embryonic development: Regulates somite formation, neural tube closure, limb and digit patterning, cardiogenesis, and pulmonary morphogenesis [1]. Cellular differentiation: A key factor in differentiation of MSC to osteoblasts, and promotes osteogenic, chondrogenic, and adipogenic pathways [1,2, 4]. Osteogenic and regenerative functions: BMP2 is a key player in skeletal physiology including Endochondral ossification: Initiates cartilage to bone transition MSC recruitment: Stimulates migration and differentiation into osteoblasts Bone matrix remodeling: Released during osteoclast-mediated resorption Fracture Healing: Promotes chondrocyte differentiation, periosteal activation, and ligament/tendon regeneration [3,4]. Tissue homeostasis: In addition to bone remodeling functions, BMP-2 also has roles in cardiac and neural tissue function throughout adulthood [1-3]. BMP-2 Signaling Pathways BMP-2 signals through serine/threonine kinase receptors (BMPRI and BMPRII), activating both Smad-dependent and non-Smad pathways: Smad-Dependent Pathway BMPRII phosphorylates BMPRI BMPRI activates Smad1/5/8 Smad1/5/8 complexes with Smad4 The complex translocates to nucleus to regulate genes like Runx2, Osx, and Dlx5 Non-Smad Pathways Activation of MAPK, ERK, and JNK cascades Crosstalk with TGF-β, Wnt, Notch, and Hedgehog pathways [1,3,5] These pathways orchestrate osteoblast differentiation, osteoclastogenesis, and overall skeletal homeostasis. Therapeutic Applications of rhBMP-2 Recombinant human BMP-2 (rhBMP-2) is widely used in clinical settings for spinal fusion surgeries, treatment of non-union fractures, and maxillofacial bone augmentation. To enhance its efficacy, rhBMP-2 is often delivered via biocompatible carriers such as collagen sponges or synthetic bone graft substitutes. Despite demonstrated osteoinductive capabilities, use of rhBMP-2 is not without challenges or safety concerns. Clinical Considerations for rhBMP-2 Use Postoperative Complications Edema & Inflammation – especially problematic in the cervical spine or facial regions Radiculitis – nerve irritation near neural structures Ectopic Bone Formation – unintended ossification outside the target site Delivery & Dosing Challenges Carrier Limitations – collagen sponges may lack mechanical support Short half-life (~1 hr): requires controlled-release systems Dosing Uncertainty: overdosing may increase complications while underdosing may result in reduced clinical efficacy Safety Concerns Carcinogenic Potential: ongoing debate with no conclusive evidence Immune Response – antibody formation is possible, usually benign Economic & Regulatory Barriers High Cost: Expensive compared to autografts/allografts Limited FDA Approval – approved for select procedures only, off-label use is common but carries risk Future Directions Understanding BMP-2 physiology is pivotal for optimizing its therapeutic potential. At Molecular Matrix, Inc., we leverage this knowledge to engineer next-generation bone graft substitutes using proprietary biomaterial technologies that support robust bone regeneration. Stay tuned for our upcoming posts, where we’ll explore innovative BMP-2 delivery systems and scaffold designs. To learn more, visit www.molecularmatrix.com. References: [1] D. Halloran, H.W. Durbano, A. Nohe, Bone Morphogenetic Protein-2 in Development and Bone Homeostasis, J. Dev. Biol. (2020) 8(3):19. doi: 10.3390/jdb8030019. [2] Q. Liu, X. Peng, X. Liu, X. Mou, Y. Guo, L. Yang, Y. Chen, Y. Zhou, Z. Shi, Z. Yang, Z. Chen, Advances in the application of bone morphogenetic proteins and their derived peptides in bone defect repair, Compos. Part B Eng. 262 (2023) 110805. https://doi.org/10.1016/j.compositesb.2023.11080. [3] L. Zhu, Y. Liu, A. Wang, Z. Zhu, Y. Li, C. Zhu, Z. Che, T. Liu, H. Liu, L. Huang, Application of BMP in Bone Tissue Engineering, Front. Bioeng. Biotechnol. 10 (2022) 810880. https://doi.org/10.3389/fbioe.2022.810880. [4] E. Segredo-Morales, P. García-García, C. Évora, A. Delgado, BMP delivery systems for bone regeneration: Healthy vs osteoporotic population. Review, J. Drug Deliv. Sci. Technol. 42 (2017) 107–118. https://doi.org/10.1016/j.jddst.2017.05.014. [5] B.-R. Keum, H.J. Kim, G.-H. Kim, D.-G. Chang, Osteobiologies for Spinal Fusion from Biological Mechanisms to Clinical Applications: A Narrative Review, Int. J. Mol. Sci. 24 (2023) 17365. https://doi.org/10.3390/ijms242417365. [6] A.V. Vasilyev, T.B. Bukharova, V.S. Kuznetsova, Yu.D. Zagoskin, S.A. Minaeva, T.E. Grigoriev, E.N. Antonov, E.O. Osidak, E.V. Galitsyna, I.I. Babichenko, S.P. Domogatsky, V.K. Popov, S.N. Chvalun, D.V. Goldshtein, A.A. Kulakov, Comparison of Impregnated Bone Morphogenetic Protein-2 Release Kinetics from Biopolymer Scaffolds, Inorg. Mater. Appl. Res. 10 (2019) 1093–1100. https://doi.org/10.1134/S2075113319050332.
Cracking the Code: How Graft Porosity and Pore Size Unlock Bone Graft Success

Bone is an extraordinary tissue: porous, self-renewing, and load-bearing; all thanks to a complex interplay of bone cells, blood vessels, and a matrix of organic
Bone Grafts and Substitutes in Fracture Repair – Part 2
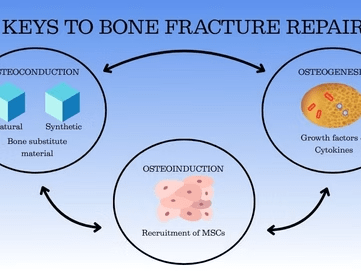
In our first post in this series, we explored the ideal characteristics of bone graft substitutes (BGS), emphasizing their osteoinductive, osteoconductive, osteogenic, bioresorbable, and biocompatible properties.
Osteoimmunology: Bridging Bone and Immune System Interactions
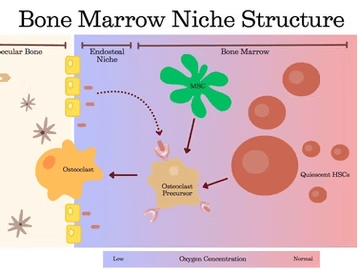
What do osteoporosis, periodontitis, and rheumatoid arthritis (RA) have in common? Could their shared mechanisms hold the key to improving bone repair?
Understanding How Autoimmunity Impacts Bones: When Your Body Can’t Tell Friend from Foe
We’ve been discussing the fascinating connections between the immune and skeletal systems in our current blog series on Osteoimmunology. But what happens when those interactions turn hostile?
Bone-Immune System Interactions: Implications for Rheumatoid Arthritis
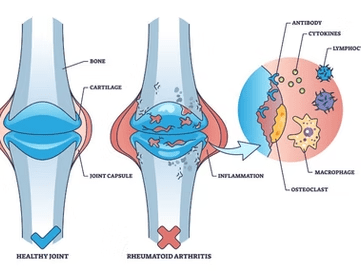
What is the most common autoimmune disorder? The correct answer is Rheumatoid Arthritis (RA), affecting more than 18 million people worldwide, with annual treatment costs estimated at $20,000 per patient.
Bone Grafts and Substitutes in Fracture Repair – Part 1
What if bones could heal faster and stronger with the help of materials designed in a lab?
From Tiny Bones to Big Breaks: A Primer on Fracture Recovery
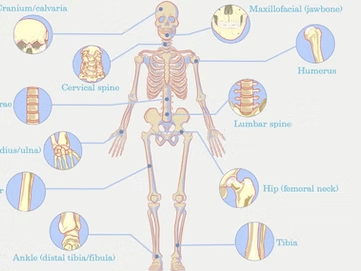
The smallest bone in the human body is roughly the size of a grain of rice! The stapes, a stirrup-shaped bone inside your middle ear, may be tiny, but it plays a huge role in hearing by transmitting sound vibrations to the inner ear.
Healing Broken Bones: The Latest Advances in Fracture Repair
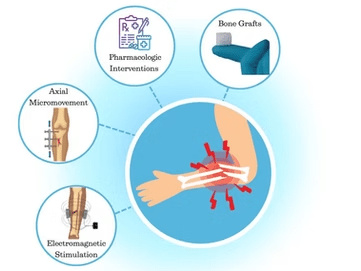
Breaking a bone is more than just an inconvenience – it’s a journey of recovery that can take weeks or even months, depending on the severity of the fracture and the treatment approach.
Harnessing Osteoimmunology to Improve Bone Health and Fracture Repair
In this blog series focused on osteoimmunology, we explored the fascinating intersection of bone biology and the immune system.
